We seek to clinically translate our engineering discoveries, and are motivated by the need for imaging biomarkers to help patients with neurological and psychiatric disorders.
Our innovations include novel MRI (magnetic resonance imaging) and PET (positron emission tomography) methods. We combine new scanning paradigms with biophysical models of brain physiology and image reconstructions to measure biomarkers that were previously not available.
The imaging biomarkers we investigate include cerebral blood flow, brain oxygenation, and cerebrovascular reactivity - how the brain's vessels respond to a "stress test". We also study the blood-brain barrier and its breakdown in neurological diseases, which alters how tracers and neuroinflammatory agents pass into the brain.
Research Areas
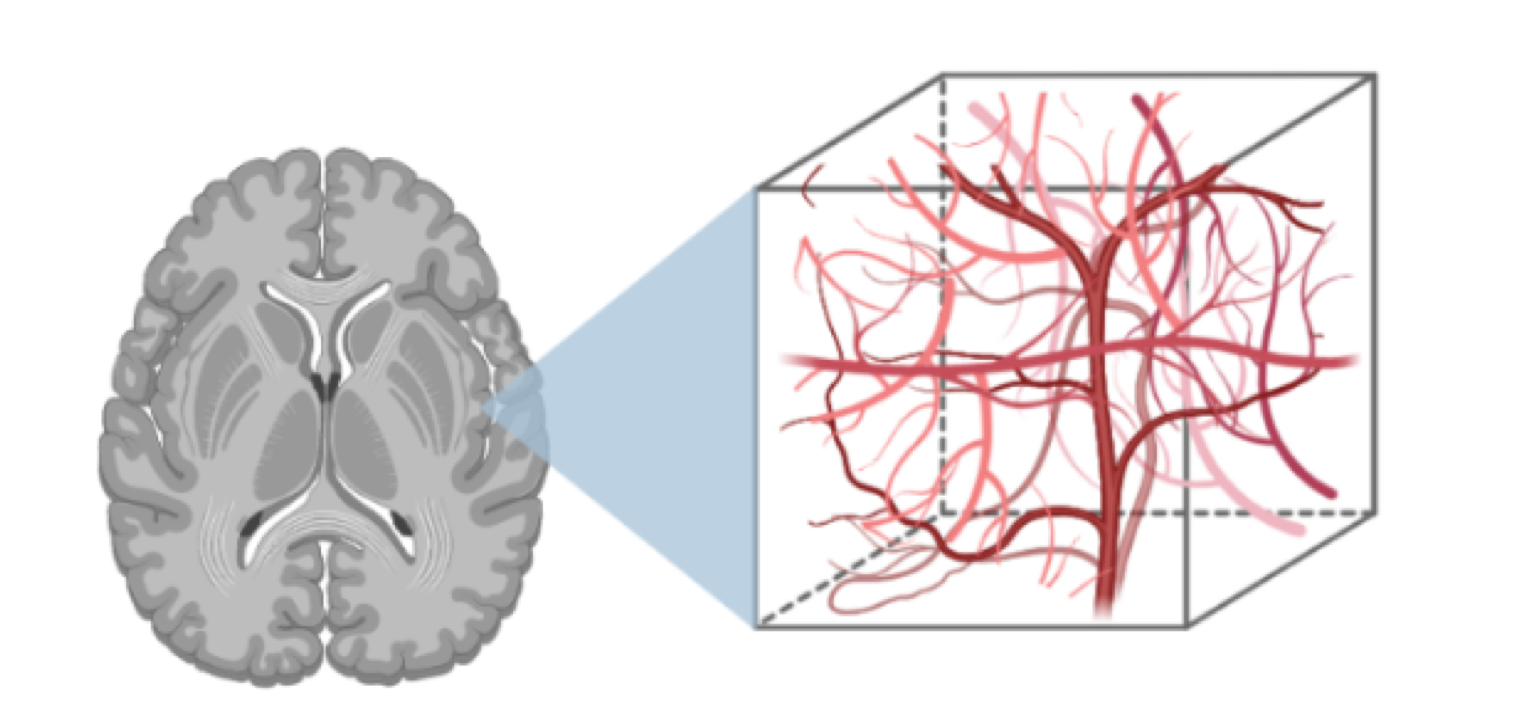
Quantitative Cerebrovascular MRI
We develop quantitative imaging tools and models to measure perfusion, brain vascular function, and reactivity. We also optimize methods to study neurofluids and the blood-brain barrier. This toolset offers a unique physiological perspective into aging / development, dementia, and psychiatric conditions.
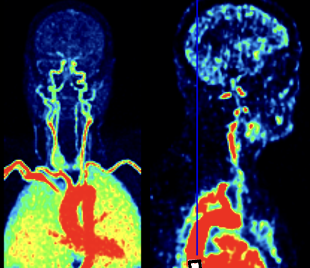
Brain PET Kinetic Modeling
Dynamic PET provides molecular sensitivity to protein accumulation and metabolism in the brain. We quantify and model PET tracer uptake in the brain to learn about its function, physiology, and metabolic nature.
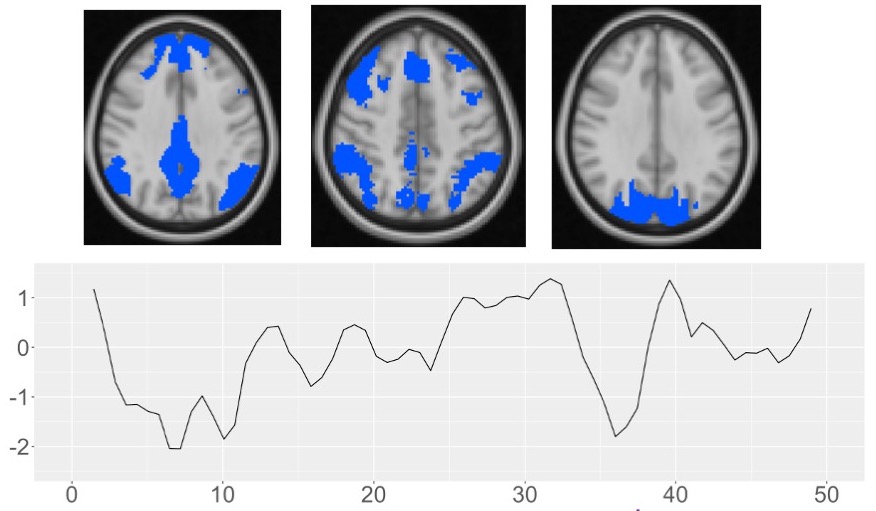
Functional Connectivity and Physiology
The brain is organized into functional networks that show correlations in the BOLD functional MRI signal, even at resting-state. We use these BOLD MRI fluctuations to learn about cerebral physiology and how functional connectivity in the brain supports learning and cognition.
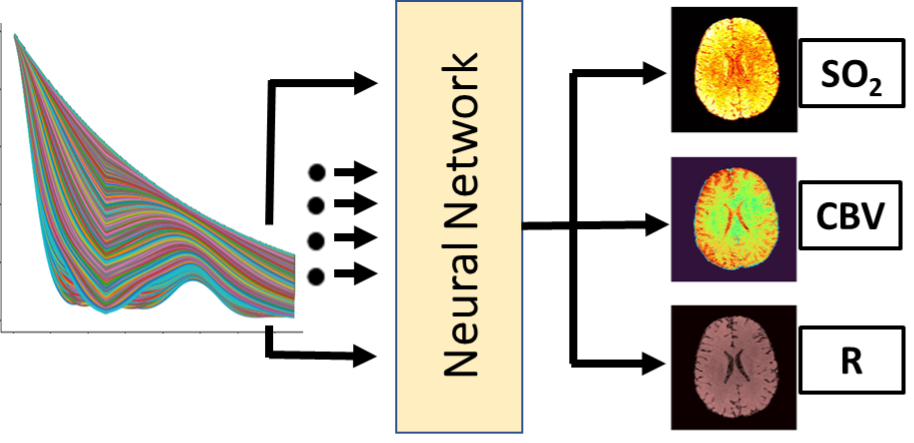
Machine Learning for Brain Mapping
Machine learning can provide exquisite opportunities for pattern matching and predictions from rich imaging information. We utilize machine learning to predict brain changes in aging and dementia and to enhance quantitative vascular MRI maps.